Trusted By Global Pharmaceutical Customers



-2.png?width=350&height=200&name=Untitled%20design%20(1)-2.png)



Pharma DSCSA Compliance Is Complex — and the Stakes Are High
Pharmaceutical manufacturers and distributors must comply with strict DSCSA regulations. Managing serialized products, reporting events, and maintaining traceability across partners can be complex and risky without the right platform.
The Drug Supply Chain Security Act requires end-to-end traceability of prescription drug products across the full distribution network — from manufacturer to dispenser. Non-compliance can result in failed FDA audits, product recalls, and significant financial penalties.
Ready to simplify DSCSA compliance?
Book a 30-minute personalized demo. No commitment required.
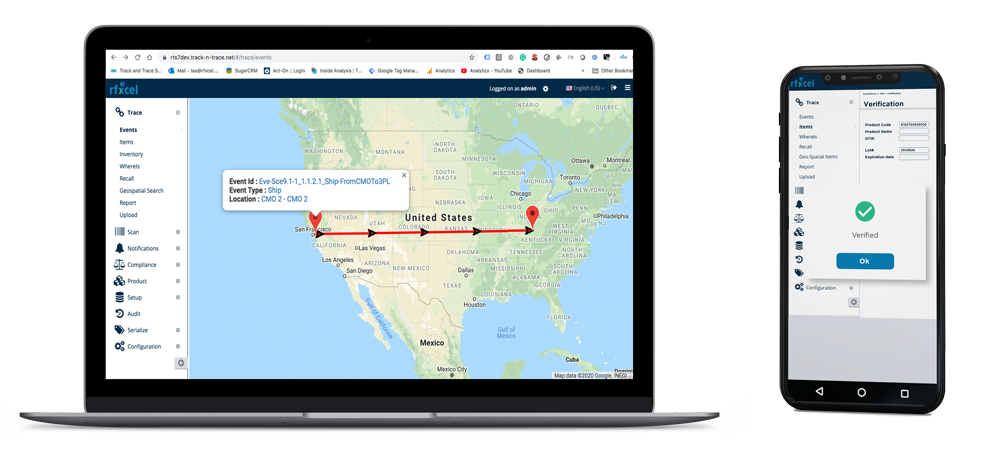
One Platform. Complete DSCSA Compliance.
Rfxcel provides a secure DSCSA compliance platform that tracks drug products across the supply chain. Our solution enables real-time serialization tracking, compliance reporting, and product verification across manufacturers, wholesalers, and distributors.
By centralizing compliance data across all supply chain partners, rfxcel eliminates the manual reconciliation and data gaps that put companies at regulatory risk.
Key benefits:
End-to-end pharmaceutical traceability
Real-time DSCSA compliance monitoring
Serialized product verification
Automated regulatory reporting
Global supply chain visibility
Everything Your DSCSA Compliance Team Needs
Serialization Tracking
Capture and report serialization events at every supply chain node — from packaging line to point of dispense — in full compliance with DSCSA requirements.
Compliance Reporting Automation
Automatically generate transaction history, transaction information, and transaction statements (T3) required under DSCSA — reducing manual effort and audit preparation time.
Secure Partner Integration
Connect with manufacturers, wholesalers, 3PLs, and dispensers using GS1, EPCIS, EDI, and API standards, and exchange serialized product data through a secure, interoperable network that meets FDA requirements.
Real-Time Product Verification
Verify the legitimacy of serialized drug products in real time at the point of receipt or dispense, reducing the risk of counterfeit or diverted products entering the supply chain.
Pharmaceutical brands trust rfxcel globally
Global regulatory markets supported out-of-the-box
Platform uptime SLA for mission-critical compliance
Days average time to full deployment
Ready to eliminate your DSCSA compliance blind spots?
Talk to a compliance specialist — free, no sales pressure.
Transform your pharmaceutical supply chain with real-time DSCSA compliance.
Join 100+ pharmaceutical companies using rfxcel to gain end-to-end supply chain visability, automate compliance, and reduce operational risk.
