Trusted By Global Pharmaceutical Customers



-2.png?width=350&height=200&name=Untitled%20design%20(1)-2.png)



Serialization Complexity Puts Compliance & Product Security at Risk
Serialization is critical for pharmaceutical compliance and product security. Without proper serialization management, companies risk regulatory violations, counterfeit drugs, and supply chain inefficiencies.
Global serialization mandates — including DSCSA in the US, EU FMD in Europe, and ANVISA in Brazil — require pharmaceutical companies to assign, track, and report unique product identifiers at every level of packaging. Managing this across multiple markets, packaging lines, and trading partners without a centralized platform creates significant operational and compliance risk.
Ready to simplify pharmaceutical serialization?
Book a 30-minute personalized demo. No commitment required.
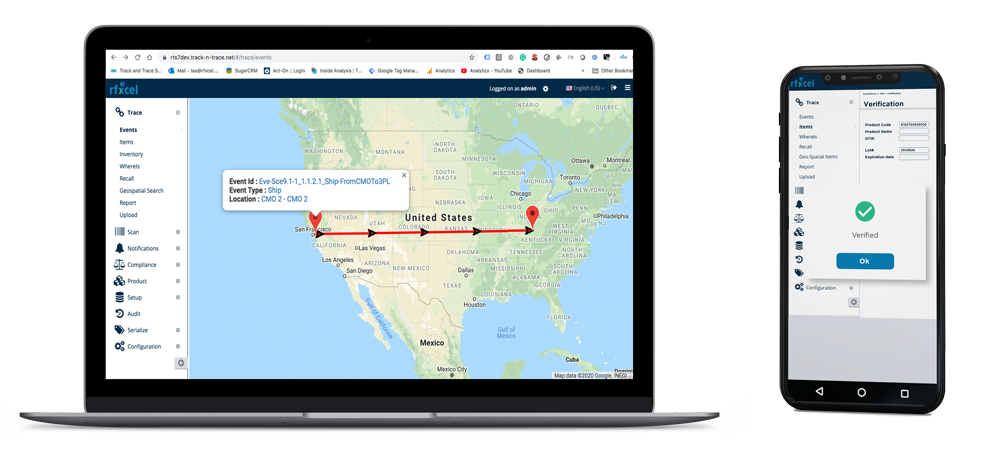
Centralized Serialization. Global Compliance.
Rfxcel serialization software helps pharmaceutical companies generate, manage, and track serialized product identifiers across packaging lines, warehouses, and distribution networks.
The platform integrates directly with packaging line equipment and connects to upstream and downstream supply chain partners — ensuring serialized data is accurate, complete, and compliant across every market you operate in.
Key benefits:
Global serialization management
Real-time serial number tracking
Packaging line integration
Regulatory compliance readiness
Built for Global Pharmaceutical Serialization
Serial Number Management
Generate, assign, and manage serial numbers and barcodes across all product lines and packaging formats, in full compliance with GS1 standards and country-specific regulatory requirements.
Aggregation Tracking
Maintain accurate parent-child relationships across item, case, and pallet levels to support full aggregation traceability throughout the supply chain.
Packaging Integration
Connect directly with existing packaging line equipment and ERP systems to capture serialization data in real time — without disrupting production operations.
Compliance Reporting
Automatically produce the compliance reports and data submissions required by DSCSA, EU FMD, and other international serialization regulations.
Pharmaceutical brands trust rfxcel globally
Global regulatory markets supported out-of-the-box
Platform uptime SLA for mission-critical compliance
Days average time to full deployment
Ready to eliminate your serialization blind spots?
Talk to a serialization specialist — free, no sales pressure.
Transform your pharmaceutical supply chain with real-time serialization.
Join 100+ pharmaceutical companies using rfxcel to gain end-to-end supply chain visibility, automate compliance, and reduce operational risk.
