Trusted By Global Pharmaceutical Customers



-2.png?width=350&height=200&name=Untitled%20design%20(1)-2.png)



Legacy Traceability Systems Are Holding Pharma Companies Back
Pharmaceutical companies must manage complex global supply chains while ensuring regulatory compliance and product integrity. Many organizations rely on legacy traceability systems that lack flexibility, visibility, and real-time data across supply chain partners. This can lead to compliance risks, operational inefficiencies, and limited visibility across the pharmaceutical distribution network.
As regulations like DSCSA tighten enforcement and global serialization mandates expand, the limitations of older platforms become increasingly costly — leaving companies struggling to keep pace with compliance requirements and partner data exchange.
Ready to see a better approach to pharma traceability?
Book a 30-minute personalized demo. No commitment required.
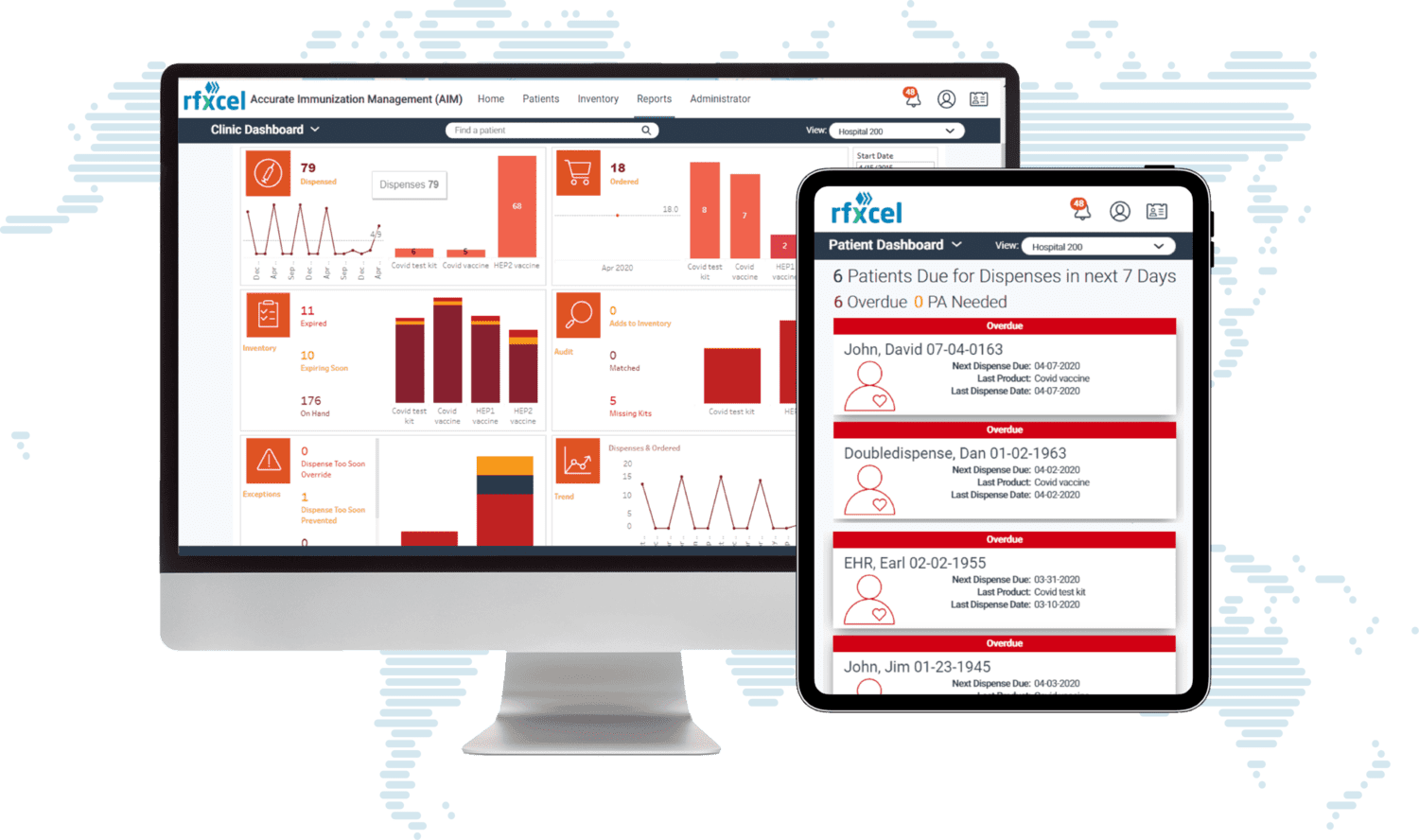
One Platform. Modern Pharmaceutical Traceability.
Rfxcel provides a modern pharmaceutical traceability platform that enables companies to monitor products, ensure regulatory compliance, and gain real-time supply chain insights. Our platform connects manufacturers, distributors, and logistics partners through a unified traceability network. This allows pharmaceutical companies to track products from manufacturing through distribution while maintaining full compliance with global regulations.
Unlike legacy systems built on rigid, outdated architectures, rfxcel is designed to scale with your business — connecting to trading partners quickly, adapting to new regulatory markets out of the box, and delivering real-time data across your entire supply chain network.
Key benefits:
Real-time pharmaceutical supply chain visibility
Secure product traceability across partners
Automated compliance monitoring and reporting
Reduced risk of counterfeit medicines
Improved supply chain efficiency
Why Pharmaceutical Companies Switch to rfxcel
Pharmaceutical manufacturers and distributors rely on rfxcel to modernize supply chain traceability and compliance operations. The platform enables organizations to gain complete supply chain visibility while maintaining strict regulatory compliance.
Supply Chain Visibility
Gain real-time insights into shipments, inventory, and logistics operations across all supply chain partners — on a single, unified platform.
End-to-End Traceability
Track pharmaceutical products across manufacturing, packaging, distribution, and pharmacy delivery — with real-time event data at every supply chain node.
Serialization Management
Manage serialized product identifiers across packaging lines and global supply chains, with built-in support for DSCSA, EU FMD, and other major regulatory frameworks.
Regulatory Compliance
Meet global pharmaceutical regulations including DSCSA requirements — with automated reporting, partner verification, and compliance monitoring built in.
Pharmaceutical brands trust rfxcel globally
Global regulatory markets supported out-of-the-box
Platform uptime SLA for mission-critical compliance
Days average time to full deployment
Ready to eliminate your traceability blind spots?
Talk to a traceability specialist — free, no sales pressure.
Transform your pharmaceutical supply chain with real-time traceability.
Join 100+ pharmaceutical companies using rfxcel to gain end-to-end supply chain visibility, automate compliance, and reduce operational risk.
